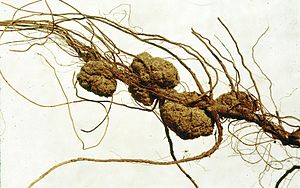
Agrobacterium tumefaciens``, dikotiledonların 140 türünden fazlasında Taç Uru hastalığına (tümör oluşumu) neden olan bir ajandır. Çubuk şeklinde gram negatif bir bacteridir (Smith ``et al.``, 1907). Semptomlar küçük bir DNA parçasının (transfer DNA olarak bilinen T-DNA) bitki hücresi içindeki bitki genomuyla yarı-rastgele bir bölgeden birleşir.
Agrobacterium, baklagillerde (simbiyont) azot fiksasyonu yapan Rhizobiaceae familyasına ait bir Alpha Proeobacterium`dur. Azot fiksasyonu yapan simbiyontların tersine tümör oluşturan Agrobacteriumlar parazittir ve bitkiye bir yarar sağlamazlar. Agrobacterium`dan etkilenen çok çeşitli bitkiler tarım endüstrisi için büyün öneme sahiptirler (Moore ``et al.``, 1997).
Ekonmik olarak ``A. tumerfaciens``, üzüm şarabı, çekirdekli meyveler ve kabuklu yemiş ağaçları için önemli patojenlerdir.
Konjugasyon
In order to be virulent, the bacterium must contain a certain plasmid, of 180kb, which encodes the T-DNA and all the genes necessary to transfer it to the plant cell. Many strains of ``A. tumefaciens`` do not contain this plasmid.Since the Ti Plasmid is essential to cause disease, pre-penetration events in the rhizosphere occur to promote conjugation - exchange of plasmids amongst bacteria. In the prescence of opines, ``A. tumerfaciens`` produces a diffusible conjugation signal called 30C8HSL or the ``Agrobacterium`` autoinducer. This activates the transcription factor TraR, positively regulating the transcription of genes required for conjugation.
Enfeksiyonun Metodu
Bağlanma
``A. tumefaciens`` have flagella that allow them to swim through the soil towards Photoassimilates that accumulate in the rhizosphere around roots. Some strains may chemostatically move towards chemicals that indicate a wounded plant cell, such as acetosyringone.
Attachement is a two step process. Following an initial weak and reversible attachement, the bacteria synthesise cellulose fibrils that anchor them to the wounded plant cell. Four main genes are involved in this process: ``chvA, chvB, pscA`` and ``att``. It appears that the products of the first three genes are involved in the actual synthesis of the cellulose fibrils. These fibrils also anchor the bacteria to each other, helping to form a microcolony.
After production of cellulose fibrils a Ca2+ dependent outer membrane protein called rhicadhesin is produced, which also aids in sticking the bacteria to the cell wall. Homologues of this protein can be found in the Rhizobium.
- Agrobacterium
a€¢ Gram negative rods a€¢ Unusual mechanism --- competitive advantage tumors in plants a€¢ `A.tumefaciens` --- Ti plasmid growth hormone and direst synthesis of opine (amino acid derivative)
T-Pilus Oluşumu
In order to tranfer the T-DNA into the plant cell ``A. tumefaceins`` uses a Type IV secretion mechanism, involving the production of a T-pilus.
The VirA/VirG two component sensor system is able to detect phenolic signals released by wounded plant cells, in particular acetosyringone. This leads to a signal transduction event activating the expression of 11 genes within the VirB operon which are responsible for the formation of the T-pilus.
First, the VirB" pro-pilin is formed. This is a polypeptide of 121 amino acids which requires processing by the removal of 47 residues to form a t-pilus subunit. The subunit is circulized by the formation of a peptide bond between the two ends of the polypeptide.
Products of the other VirB genes are used to transfer the subunits across the plasma membrane. Yeast Two-hybrid studies provide evidence that VirB6, VirB7, VirB8, VirB9 and VirB10 may all encode components of the transporter. An ATPase for the active transport of the subunits would also be required.
T-DNA`nın Bitki Hücresi İçine Taşınması
The T-DNA must be cut out of the circular plasmid. A VirD1/D2 complex causes nicks in the DNA at the left and right borders. A single stranded DNA binding protein, VirE2, then coats the T-DNA and a VirD2 protein is covalently attached to the 5` end. VirD2 contains a motif that leads to the nucleoprotein complex being targeted to the type IV secretion system.
Once inside the cell, nuclear localization signals, or NLS, located on the VirE2 and VirD2 are recognised by the importin alpha protein, which then associates with importin beta and the nuclear pore complex to transfer the T-DNA into the nucleus. VIP1 also appears to be an important protein in the process, possibly acting as an adapter to bring the VirE2 to the importin. Once inside the nucleus, VIP2 may target the T-DNA to areas of chromatin that are being actively transcribed, so that the T-DNA can integrate into the host genome.
T-DNA Üzerindeki Genler
Hormonlar
In order to cause gall formation the T-DNA encodes genes for the production of auxin or indole-3-acetic acid via the IAM pathway. This biosynthetic pathway is not used in many plants for the production of auxin, so it means the plant has no molecular means of regulating it and auxin will be produced constitutively. Genes for the production of cytokinins are also expressed. This stimulates cell proliferation and gall formation.
Opinler
The T-DNA contains genes encoding enzymes that cause the plant to create specialized amino acids which the bacteria can metabolize, called opines (Zupan ``et al.``, 2000). Opines are a class of chemicals that serve as a source of energy for A. tumefaciens, but not for most other organisms. The specific type of opine produced by A. tumefaciens C58 infected plants is nopaline (Escobar ``et al.``, 2003).
Two nopaline type Ti plasmids, pTi-SAKURA and pTiC58, were fully sequenced. Agrobacterium tumefaciens C58, the first fully sequenced pathovar, was first isolated from a cherry tree crown gall. The genome was simultaneously sequenced by Goodner ``et al.``, 2001 and Wood ``et al.``, 2001. The genome of A. tumefaciens C58 consists of a circular chromosome, two plasmids, and a linear chromosome. The presence of a covalently bonded circular chromosome is common to Bacteria, with few exceptions. However, the presence of both a single circular chromosome and single linear chromosome is unique to a group in this genus. The two plasmids are pTiC58, responsible for the processes involved in virulence, and pAtC58, coined the “cryptic” plasmid (Goodner ``et al.``, 2001) (Wood ``et al.``, 2001).
The pAtC58 plasmid has been shown to be involved in the metabolism of opines and to conjugate with other bacteria in the absence of the pTiC58 plasmid (Vaudequin-Dransart ``et al.``, 1998). If the pTi plasmid is removed the tumor growth that is the means of classifying this species of bacteria does not occur.
Yararlı Kullanım Alanları
The DNA transmission capabilities of ``Agrobacterium`` have been extensively exploited in biotechnology as a means of inserting foreign genes into plants. Marc Van Montagu and Jeff Schell, (University of Ghent and Plant Genetic Systems, Belgium) discovered the gene transfer mechanism between Agrobacterium and plants, which resulted in the development of methods to alter Agrobacterium into an efficient delivery system for gene engineering in plants (Schell J, Van Montagu M., 1977). The plasmid T-DNA that is transferred to the plant is an ideal vehicle for genetic engineering (Zambryski, 1983). This is done by cloning a desired gene sequence into the T-DNA that will be inserted into the host DNA. This process has been performed using firefly luciferase gene to produce glowing plants. This luminescence has been a useful device in the study of plant chloroplast function and as a reporter gene (Root, 1988). Under laboratory conditions the T-DNA has also been transferred to human cells, demonstrating the diversity of insertion application (Kunik ``et al.``, 2001).The mechanism by which ``Agrobacterium`` inserts materials into the host cell by a type IV secretion system, is very similar to mechanisms used by pathogens to insert materials (usually proteins) into human cells by type III secretion. It also employes a type of signaling conserved in many Gram-negative bacteria called quorum sensing. This makes ``Agrobacterium`` an important topic of medical research as well.
Kaynaklar
- Goodner B, Hinkle G, Gattung S, Miller N, ``et al.`` 2001. Genome Sequence of the Plant Pathogen and Biotechnology Agent Agrobacterium tumefaciens C58. Science. 294:2323-2328.
- Kunik T, Tzfira T, Kapulnik Y, Gafni Y, Dingwall C, Citovsky V. 2001. Genetic transformation of HeLa cells by Agrobacterium. Proc. Natl. Acad. Sci. 98:1871-1876.
- Moore LW, Chilton WS, Canfield ML. 1997. Diversity of Opines and Opine-Catabolizing Bacteria Isolated from Naturally Occurring Crown Gall Tumors. App. Environ. Microbiol. 63:201-207.
- Root M. 1988. Glow in the dark biotechnology. Bioscience. 38:745-747.
- Schell J, Van Montagu M., The Ti-plasmid of Agrobacterium tumefaciens, a natural vector for the introduction of nif genes in plants?, Basic Life Sci. 1977;9:159-79.
- Vaudequin-Dransart V, Petit A, Chilton WS, Dessaux Y. 1998. The cryptic plasmid of Agrobacterium tumefaciens cointegrates with the Ti plasmid and cooperates for opine degradation. Molec. Plant-microbe Interact. 11:583-591.
- Wood DW, Setubal JC, Kaul R, Monks DE, ``et al.`` 2001. The Genome of the Natural Genetic Engineer Agrobacterium tumefaciens C58. Science. 294:2317-2323.
- Zambryski P. ``et al.`` 1983. Ti plasmid vector for introduction of DNA into plant cells without alteration of their normal regeneration capacity. EMBO J. 2:2143-2150.
- Zupan J, Muth TR, Draper O, Zambryski P. 2000. The transfer of DNA from Agrobacterium tumefaciens into plants: a feast of fundamental insights. Plant J. 23:11-28.
- Dikinson, M 2003. Molecular Plant Pathology. Published by BIOS Scientific publishers.
- Lal, Erh-Min and Kado, Clarence I. 2000. The T-Pilus of ``Agrobacterium tumefaciens``. Trends in Microbiology, Vol. 8, Issue 8.
- Ward, Doyle V., Zupan, John R and Zambryski, Patricia C. 2002. ``Agrobacterium`` VirE2 gtes the VIP1 treatment in plant nuclear import. Trends in Plant Science, Vol. 7 Issue 1.
Linkler
- ``Agrobacterium`` tumefaciens C58 Genome Page - Sequenced by Cereon Genomics/University of Richmond
- ``Agrobacterium`` tumefaciens C58 Genome Page - Sequenced by DuPont/Univ of Washington/Univ of Campinas
Bitki hastalıkları